My Projects
Development of Bioengineered Brain Targeting Approaches
My interdisciplinary research unites two strands of most recent crucial areas of neuroscience; development of bioengineered brain targeting approaches to improve the lives of millions of patients suffering from neurodegeneration, stroke, and other brain disorders. In response to these challenges herein, I designed novel mussel inspired polydopamine (PDA)-coated mesoporous silica nanoparticles (PDA-AMSNs) with excellent antioxidative ability to deliver a new potential therapeutic GSK-3β inhibitor lead small molecule abbreviated as Neuro Chemical Modulator (NCM) at the TBI site using a neuroprotective peptide hydrogel (PANAP). PDA-AMSNs loaded with NCM (i.e., PDA-AMSN-D) into the matrix of PANAP were injected into the damaged area in an in vivo cryogenic brain injury model (CBI). This approach is specifically built while keeping the logic AND gate circuit as the primary focus. Where NCM and PDA-AMSNs act as two input signals and neurological functional recovery as a single output. Therapeutically, PDA-AMSN-D significantly decreased infarct volume, enhanced neurogenesis, rejuvenated BBB senescence, and accelerated neurological function recovery in a CBI.
Garg et al., ACS Appl. Mater. Interfaces 2024, 16, 28, 36168–36193
ECM mimicking peptide based materials for brain injury and enhanced neurogenesis
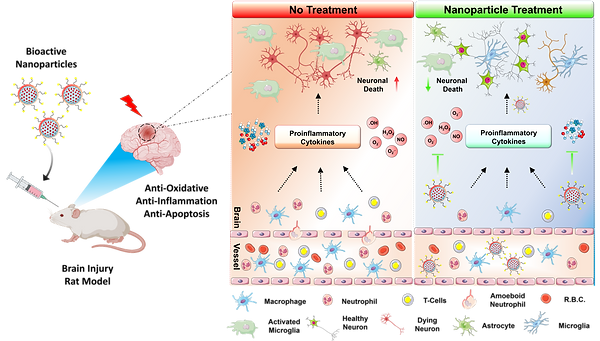
Traumatic brain injuries (TBIs) causes multifaceted disruption in neural network, initiate huge inflammation process and formed glial scars resulted severe damage to brain. Towards this aim, I hereby developed an extremely effective therapeutic strategy. This interesting approach showcased development and validation of a combination therapy comprising of a neuro-regenerative protective peptide hydrogel (SLNAP) and a potent neuro-regenerative chemical modulator (NCM). We found that this combination therapeutic strategy is highly effective in in vivo brain injury model establishes that this could be potential and highly effective therapeutic strategy for TBI.
Attenuates LPS Induced Neuroinflammation via TLR-4, Nrf-2 / Keap-1 Signaling Pathway
Mitochondria are complex organelle that plays a pivotal role in energy metabolism, regulation of stress responses, and also serve as a major hub for biosynthetic processes. In addition to their well-established function in cellular energetics, it also serves as the primary site for the origin of intracellular reactive oxygen species (ROS), which function as signaling molecules and can lead to oxidative stress when generated in excess. Moreover, mitochondrial dysfunction is one of the leading cause of neuroinflammation. In this regard we have rationally designed a triazine derived mitochondriotropic antioxidants (Mito-TBA), based on gallic acid and triphenylphosphonium (TPP) cation to specifically target mitochondria to mitigate neuroinflammation. In vitro Mito-TBA-3 inhibits mitoautophagy, offers neuroprotection by inhibiting the LPS induced TLR-4 activation and activating the Nrf-2/ ARE pathway in PC-12 derived neurons. In vivo Mito-TBA-3 rescue memory deficit, reversed depression like behavior, inhibit neuroinflammation, decrease proinflammatory cytokines in LPS induced neuroinflammation rat model. Overall, based on biophysical, in vitro and in vivo analysis, Mito-TBA-3 offers valuable insights as a potent therapeutic lead molecule to combat neurodegeneration even outperforming a well-known non-steroidal anti-inflammatory drug (Aspirin), it also has the potential to use as a promising therapeutic candidate for other mitochondrial oxidative stress related disorders.

Garg et al., Free Radical Biology and Medicine (Under Review)